Scheme 4-26. Ir/dtbpy-catalyzed borylation of quinolone 4-8 with B2pin2... | Download Scientific Diagram
![B2pin2-catalyzed oxidative cleavage of a C [[double bond, length as m-dash]] C double bond with molecular oxygen - Organic Chemistry Frontiers (RSC Publishing) B2pin2-catalyzed oxidative cleavage of a C [[double bond, length as m-dash]] C double bond with molecular oxygen - Organic Chemistry Frontiers (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C8QO01412D)
B2pin2-catalyzed oxidative cleavage of a C [[double bond, length as m-dash]] C double bond with molecular oxygen - Organic Chemistry Frontiers (RSC Publishing)

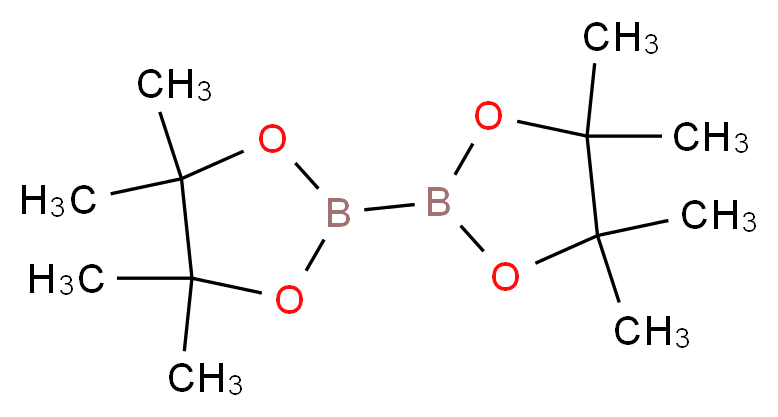
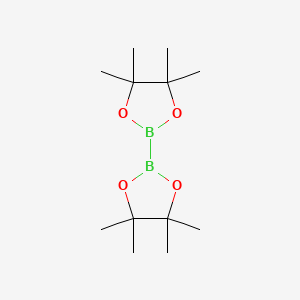
73183-34-3 | Bis(pinacolato)diboron | 2-(4,4,5,5-Tetramethyl-1,3,2 -dioxaborolan-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane; 4,4,4',4',5,5,5',5'-Octamethyl-2,2'-bis(1,3,2-dioxaborolane); 4,4',5,5'-Octamethyl-2,2'-bi-1,3,2-dioxaborolane; B2Pin2; Bis ...

Base-Catalyzed Borylation/B–O Elimination of Propynols and B2pin2 Delivering Tetrasubstituted Alkenylboronates | Organic Letters

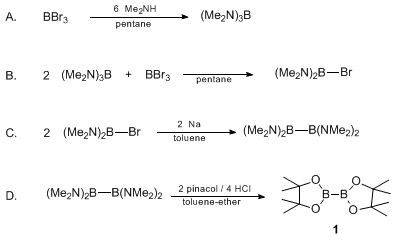
B–B bond activation and NHC ring-expansion reactions of diboron(4) compounds, and accurate molecular structures of B2(NMe2)4, B2eg2, B2neop2 and B2pin2 - Dalton Transactions (RSC Publishing)
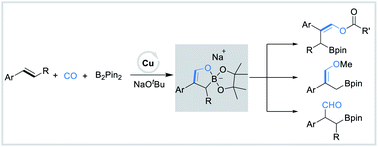
Copper-catalyzed borofunctionalization of styrenes with B2pin2 and CO - Chemical Science (RSC Publishing)

B2pin2-mediated copper-catalyzed oxidation of alkynes into 1,2-diketones using molecular oxygen - ScienceDirect

Reaction scheme of methane borylation with B2pin2 The molecular sizes... | Download Scientific Diagram
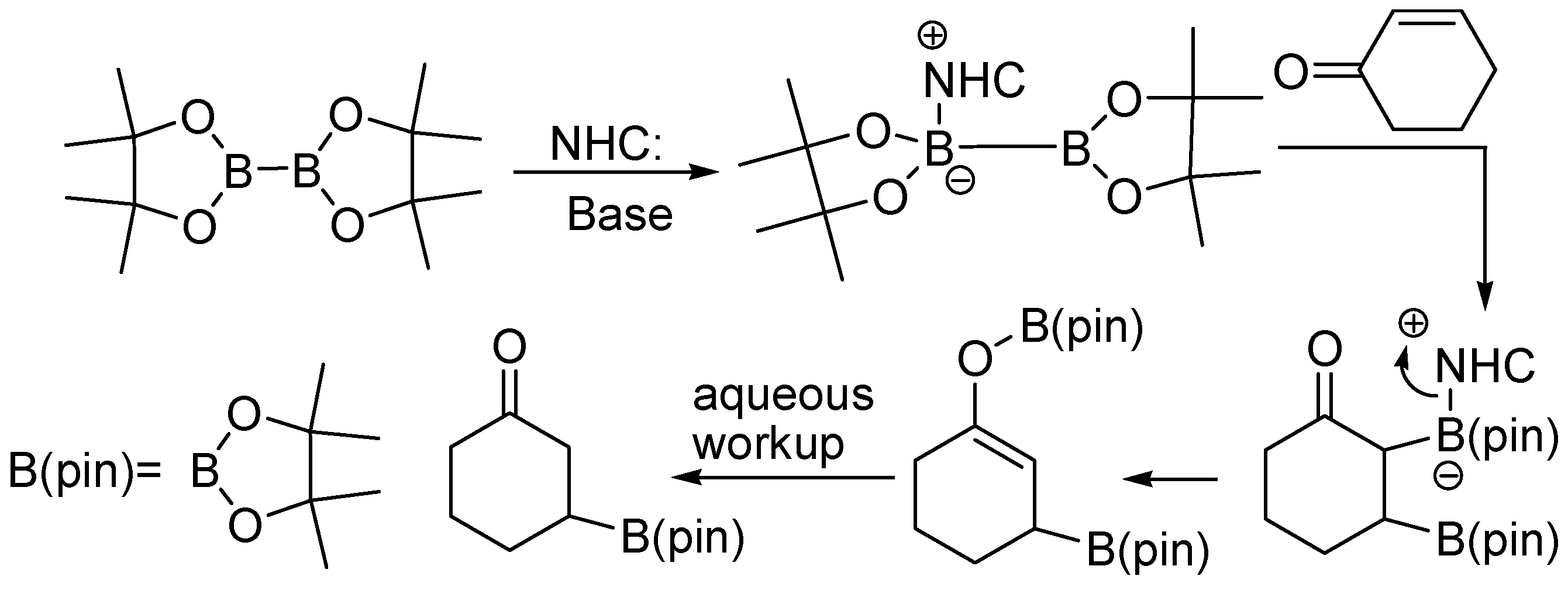
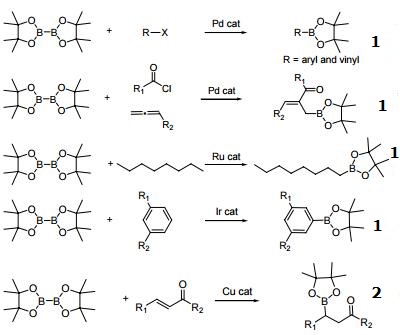
Molecules | Free Full-Text | Recent Synthesis Developments of Organoboron Compounds via Metal-Free Catalytic Borylation of Alkynes and Alkenes

B2pin2-mediated copper-catalyzed oxidation of alkynes into 1,2-diketones using molecular oxygen - ScienceDirect

Nanomaterials | Free Full-Text | α,β-Enone Borylation by Bis(Pinacolato)Diboron Catalyzed by Cu3(BTC)2 Using Cesium Carbonate as a Base

توییتر \ KingDraw در توییتر: «#namereaction The Miyaura borylation reaction enables the synthesis of boronates by cross-coupling of bis(pinacolato)diboron (B2pin2) with aryl halides and vinyl halides. #KingDraw #chemistry #science https://t.co/s8WZtTTJLC»

CuII and Cu0 Catalyzed Mono Borylation of Unsaturated Hydrocarbons with B2pin2: Entering into the Water - Stavber - 2014 - ChemCatChem - Wiley Online Library

Scheme 2. Kinetic isotope effect study. Reaction conditions: i) B2pin2... | Download Scientific Diagram